
Microorganisms | Free Full-Text | TonB-Dependent Transporters in Sphingomonads: Unraveling Their Distribution and Function in Environmental Adaptation | HTML

Reconstitution of bacterial outer membrane TonB-dependent transporters in planar lipid bilayer membranes | PNAS

TonB or not TonB: is that the question? This paper is one of a selection of papers published in a Special Issue entitled CSBMCB 53rd Annual Meeting — Membrane Proteins in Health

Substrate-induced exposure of an energy-coupling motif of a membrane transporter | Nature Structural & Molecular Biology

New substrates for TonB-dependent transport: do we only see the 'tip of the iceberg'?: Trends in Biochemical Sciences
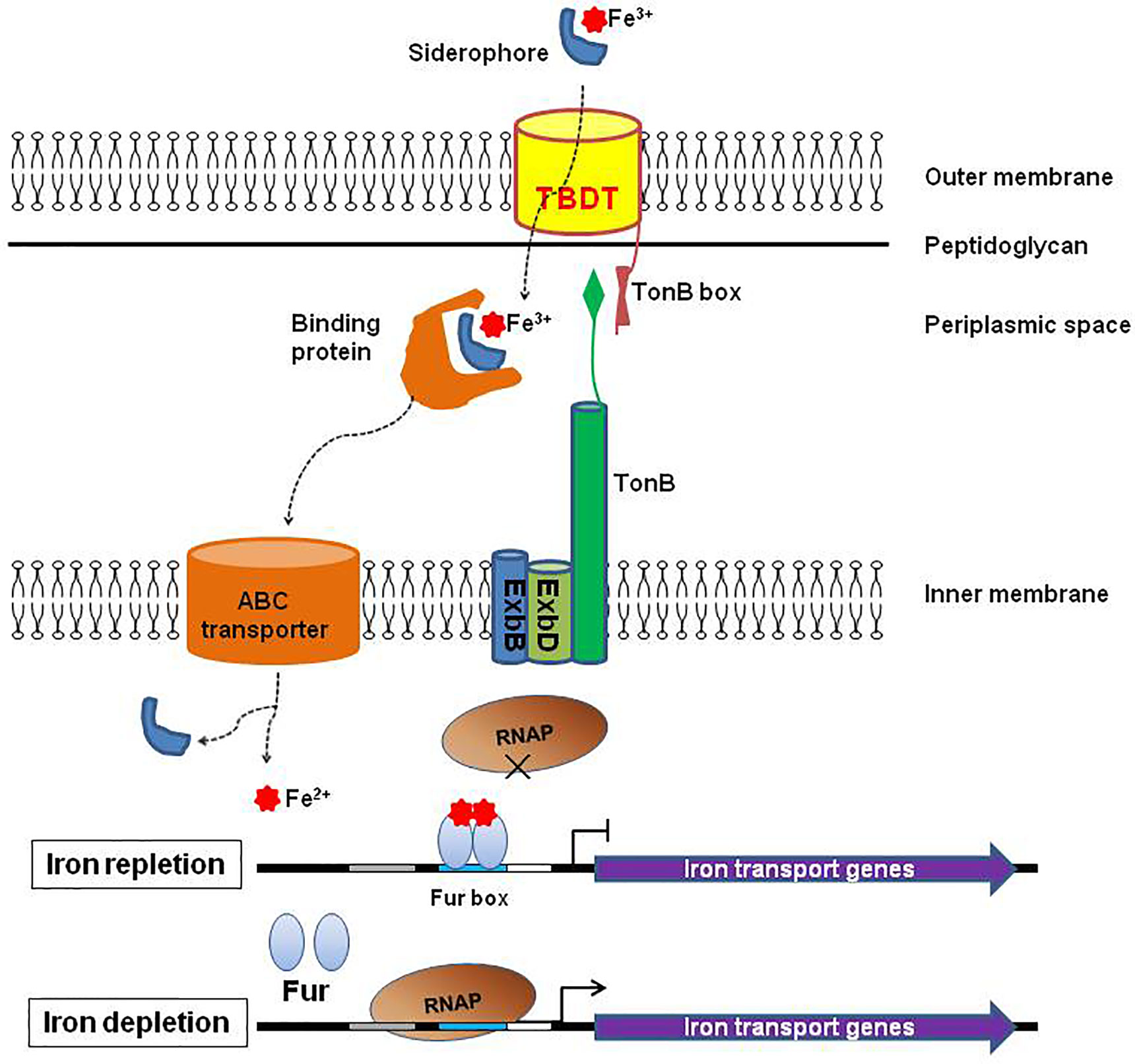
Frontiers | Application of TonB-Dependent Transporters in Vaccine Development of Gram-Negative Bacteria | Cellular and Infection Microbiology

Interactions between the Outer Membrane Ferric Citrate Transporter FecA and TonB: Studies of the FecA TonB Box | Journal of Bacteriology
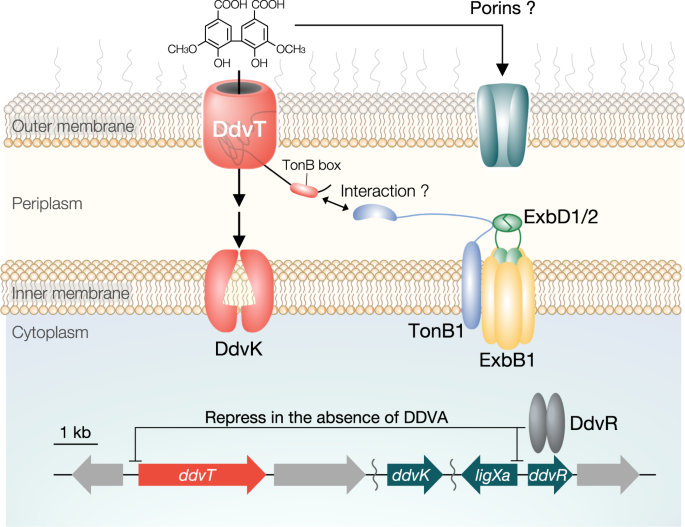
A TonB-dependent receptor constitutes the outer membrane transport system for a lignin-derived aromatic compound | Communications Biology

Interactions of the FhuA Ton box with the C-terminal domain of TonB.... | Download Scientific Diagram

New substrates for TonB-dependent transport: do we only see the 'tip of the iceberg'? - ScienceDirect

Predicting the Complex Structure and Functional Motions of the Outer Membrane Transporter and Signal Transducer FecA: Biophysical Journal

Monomeric TonB and the Ton box are required for the formation of a high-affinity transporter-TonB complex. | Semantic Scholar
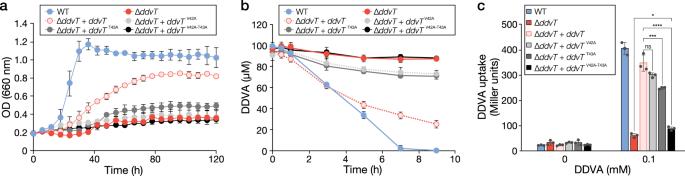
A TonB-dependent receptor constitutes the outer membrane transport system for a lignin-derived aromatic compound | Communications Biology

TonB or not TonB: is that the question? This paper is one of a selection of papers published in a Special Issue entitled CSBMCB 53rd Annual Meeting — Membrane Proteins in Health

TonB or not TonB: is that the question? This paper is one of a selection of papers published in a Special Issue entitled CSBMCB 53rd Annual Meeting — Membrane Proteins in Health

Site-directed disulfide bonding reveals an interaction site between energy-coupling protein TonB and BtuB, the outer membrane cobalamin transporter | PNAS
The TonB box. (A) Sequence alignments of TonB boxes. Shown are sequence... | Download Scientific Diagram

Gating of TonB-dependent transporters by substrate-specific forced remodelling | Nature Communications

Ternary structure of the outer membrane transporter FoxA with resolved signalling domain provides insights into TonB-mediated siderophore uptake | eLife
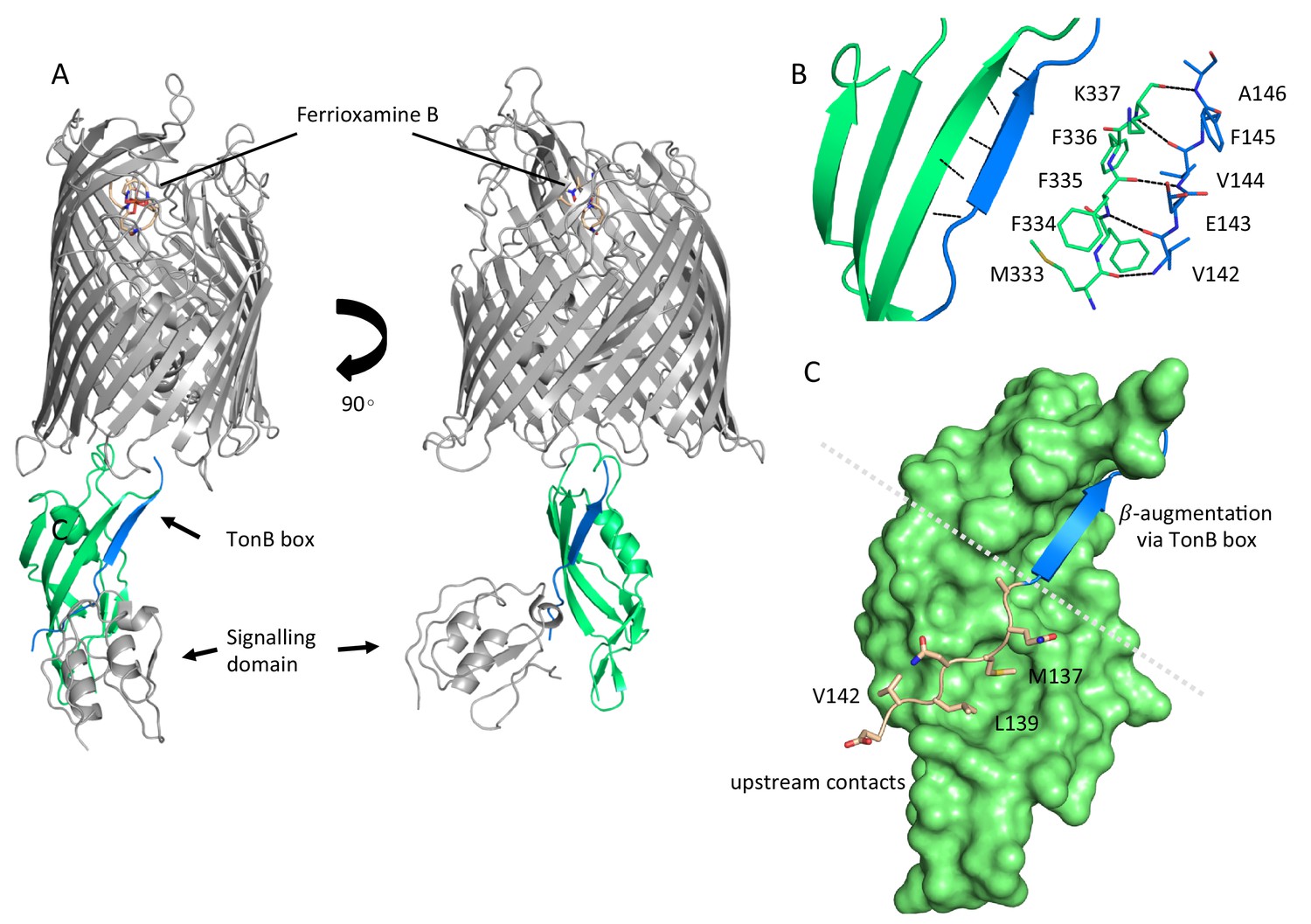



![NMR structure of the C-terminal domain of TonB protein from Pseudomonas aeruginosa [PeerJ] NMR structure of the C-terminal domain of TonB protein from Pseudomonas aeruginosa [PeerJ]](https://dfzljdn9uc3pi.cloudfront.net/2018/5412/1/fig-6-2x.jpg)

